- Seeking Series B investment for next phase of clinical trials
Background
OVM is a clinical-stage biopharma developing advanced therapeutic cancer vaccines

Immunotherapy is effective by activating the immune system to attack tumours directly thus reducing the need for invasive treatments such as chemotherapy and radiotherapy.
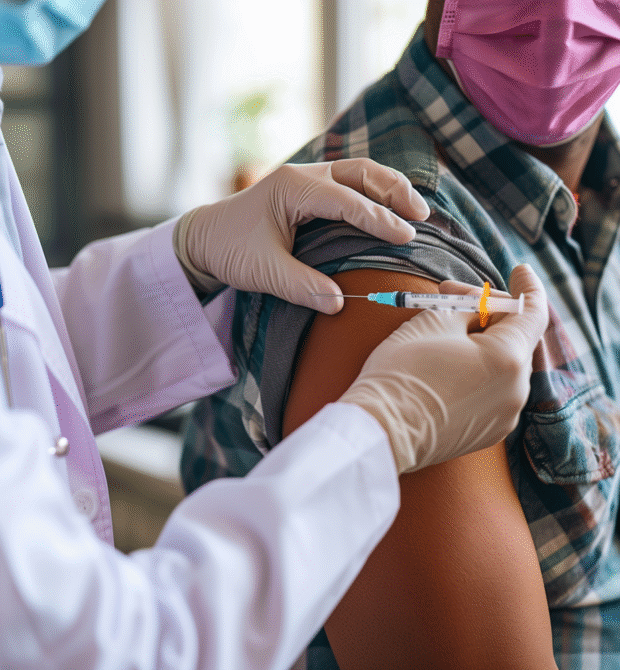
Redefining personalised medicine

Recombinant Overlapping Peptides (ROP)

Focus on novel cancer therapeutics

Patents: Strong proprietary position
Trust Towards OurnShared Purpose

Empowered To Change,nAct, And To Adapt

New AdvancednInstruments
Our Management Team



Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.


Company Timeline
Trial extended to include additional immunisations.
Announces Lead investment of $3 million in Oxford from existing investors. Prostate Cancer Research invests in Series B.
The dose escalation and safety part of the clinical trial was completed with no serious adverse events (SAEs).
Oxford Vacmedix announces Innovate UK funding for development of novel T-cell potency assays, supporting the future development of immunotherapy and vaccines.
First patient treated in OVM-200 clinical trial. The Clinical Trial begins with the treatment of the first patient at the Sarah Cannon Research Institute in London.
MHRA approval of Phase 1 clinical trial of OVM-200. Regulatory approval of OVM-200 for Phase 1 signals a significant step for Oxford Vacmedix with the first clinical trial of a cancer vaccine based on ROP technology.
OVM starts pre-clinical development in earnest following Series A investment of $12.5m USD.
With an exclusive licence to develop ROP technology, developed by Professor Shisong Jiang, from the University of Oxford.




